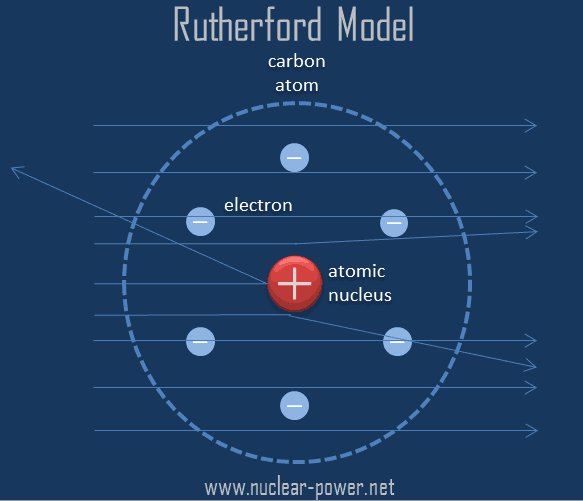
Through his experiments on radioactivity and the. His groundbreaking discoveries revolutionized our knowledge of atomic theory, paving the way for advancements in the field. atomic theory, ancient philosophical speculation that all things can be accounted for by innumerable combinations of hard, small, indivisible particles (called atoms) of various sizes but of the same basic material or the modern scientific theory of matter according to which the chemical elements that combine to form the great variety of. The Rutherford atomic model does not account for the initial energy and subsequent energy level changes. Ernest Rutherford’s contributions to the understanding of atomic structure and the elements have had a significant impact on the field of chemistry and the periodic table. They give off electromagnetic radiation due to the circular motion of orbiting thus they must have some initial energy by the law of conservation of energy. Electrons increase and decrease energy levels randomly due to the acceleration and are not always in a standard circular orbit.Electromagnetic radiation from the electrons in orbit would cause the atom to collapse into the nucleus in 10 -8 seconds. Rutherford proposed that electrons orbit around the nucleus in set paths, but according to Maxwell’s theory, this is not possible because the atom would not be stable.The model is missing parts and does not account for the location or distribution of electrons.Briefly describe the contributions made to atomic theory by the following scientist: E. d) He discovered the existence of a center to the atom. 108) deduced from this that the atom should consist of a heavy central mass. What was Ernest Rutherfords contribution to our understanding of atomic structure a) He discovered the mass of the electron. While common models today are based on the Rutherford atomic theory, it does not paint the complete picture: model of the atom and Rutherford (1911 reprinted in this volume on p. While most alpha particles passed through, a few were deflected by the nucleus, proving that there was a small, densely packed positive mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
